 It is a very ductile, pale metal, which darkens in the presence of trace amounts of oxygen. Of all elements that are solid at room temperature, caesium is the softest: it has a hardness of 0.2 Mohs. Nonradioactive caesium compounds are only mildly toxic, but the pure metal's tendency to react explosively with water means that caesium is considered a hazardous material, and the radioisotopes present a significant health and environmental hazard.Ĭharacteristics Physical properties High-purity caesium-133 stored in argon. The radioactive isotope caesium-137 has a half-life of about 30 years and is used in medical applications, industrial gauges, and hydrology. Since the 1990s, the largest application of the element has been as caesium formate for drilling fluids, but it has a range of applications in the production of electricity, in electronics, and in chemistry. Since then, caesium has been widely used in highly accurate atomic clocks. In 1967, acting on Einstein's proof that the speed of light is the most-constant dimension in the universe, the International System of Units used two specific wave counts from an emission spectrum of caesium-133 to co-define the second and the metre. The first small-scale applications for caesium were as a " getter" in vacuum tubes and in photoelectric cells. The German chemist Robert Bunsen and physicist Gustav Kirchhoff discovered caesium in 1860 by the newly developed method of flame spectroscopy. It has the largest atomic radius of all elements whose radii have been measured or calculated, at about 260 picometers. Caesium-137, a fission product, is extracted from waste produced by nuclear reactors. It has only one stable isotope, caesium-133. It is the least electronegative element, with a value of 0.79 on the Pauling scale. It is pyrophoric and reacts with water even at −116 ☌ (−177 ☏). Caesium has physical and chemical properties similar to those of rubidium and potassium. It is a soft, silvery-golden alkali metal with a melting point of 28.5 ☌ (83.3 ☏), which makes it one of only five elemental metals that are liquid at or near room temperature. Saulsġ998.Caesium ( IUPAC spelling cesium in American English) is a chemical element with the symbol Cs and atomic number 55. Structure of Crystals Crystal Lattices Unit Cells From Unit Cell to Lattice From Lattice to Unit Cell Stoichiometry Packing & Geometry Simple Cubic Metals Close Packed Structures Body Centered Cubic CesiumĬhloride Sodium Chloride Rhenium Oxide Niobium Oxide Except as otherwise noted, all images, moviesīy Dr. We may also consider the neighborhood in which each ion finds itself.Įach Cs + is surrounded by 8 Cl - (so the Cs +Ĭoordination number is 8) at the corners of each cube.Įach Cl - is also surrounded by 8 Cs + at theĬorners of a cube, so the Cl - has CN = 8. This is obvious if we compare the CsCl unit cell with the simpleĬubic unit cell showing the interstitial site. Taking a simple cubic Cs lattice and placing Cl into the interstitial sites. We can therefore think of making the CsCl by Recall that the simple cubic lattice has large interstitial sitesīetween each 8 atoms. (The two sublattices are not necessarily the same, although in this We can describe the CsCl structure as two interpenetrating simple cubic (the Cs sublattice), and only the gold Cl - (the Cl sublattice). This animation shows the CsCl lattice, only the teal Cs + Ignoring the Cs +, we note that the Cl - themselvesĬonsidering only the Cs +, they form a simple cubic We begin with the larger (gold colored) Cl - ions. One way to describe the crystal is to consider the cations and anions Click on the images below to view the open structure rotating. The same structure, but with the ions moved further apart allows However, the tightly-packed structures make it difficult to Notice that you cannot see through the structure there are When these unit cells are stacked into a lattice, theyĬlick on the images below to view the cesium lattice Click on the unit cell above to view a movie of the unitĬell rotating. One simple ionic structure is: Cesium Chloride Cesium chloride crystallizes in a cubic lattice. 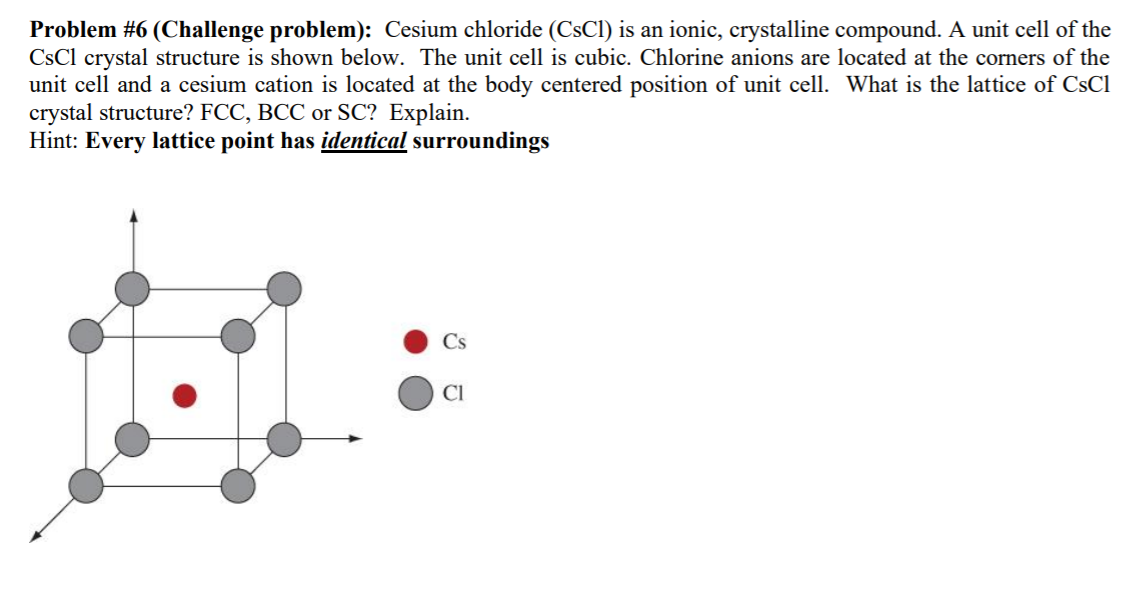
They will thus pack differently in different nitrate, carbonate, azide)Īre very non-spherical in shape. The structure must balance both types of forces. (2) The cations attract the anions, but like In the lattice, generally of different sizes. (1) There are now at least two kinds of particles Ionic compounds generally have more complicated Cesium Chloride Crystal Lattice CESIUM CHLORIDE AN IONIC COMPOUND 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
